Implant indications
Titanium vs Zirconia
Introduction
In the current literature, there is still scarce documentation on zirconia implants. Some authors have compared it to that of titanium implants fifteen years ago.
At the moment, we cannot definitively state that zirconia implants are going to become a reliable metal-free implant alternative. Yet patients are becoming increasingly interested in metal-free prosthetic treatments. For this reason, clinicians should be ready for a scenario where they will have to manage cases involving zirconia implants.
“Patients are becoming increasingly interested in metal-free prosthetic treatments”
People who are ‘Pro Zirconia’ and defend its use claim that zirconia implants can now be considered among our ‘medical arsenal’ to treat patients who demand non-metallic implants.
Pro titanium
The speaker said that a common treatment option is to use titanium implants and ceramic abutments/reconstructions. The speaker explained that since he prefers a two-piece bone-level design, he uses titanium implants because there is still insufficient documentation on two-piece bone-level zirconia implants.
“A common treatment option is to use titanium implants and ceramic abutments/reconstructions”
Regarding plaque accumulation and soft tissue response: current research shows conflicting results with respect to titanium. Although the aesthetic appearance seems to be one of the main benefits and ‘strong points’ of zirconia, the use of ceramic reconstructions on titanium implants seems to work quite well in terms of aesthetics.
Titanium implants have been demonstrated to be highly predictable in the long term. The survival of implants supporting single crowns was recently evaluated in two meta-analyses (Jung et al. 2012; Rabel et al. 2018). The results at ten years showed survival rates of 95–97%.
“But titanium implants also face several disadvantages”
But titanium implants also face several disadvantages. An increasing number of cases involving peri-implant recessions leading to mucosal discolorations have been reported (Buser et al. 2017). Another potential problem is the presence of titanium particles as a result of tribocorrosion, which may elicit a pro-inflammatory response or hypersensitivity (Mombelli et al. 2018). Until now, no pathology has been associated with the presence of titanium particles in peri-implant tissues. Out of millions of implant patients, only a few case histories have presented relatively strong evidence of titanium allergies (Albrektsson et al. 2018). It should be noted that zirconia particles have also been found in tissues surrounding zirconia implants, and the potential biological effects of this are not yet known.
Presentation figures

Figure 1
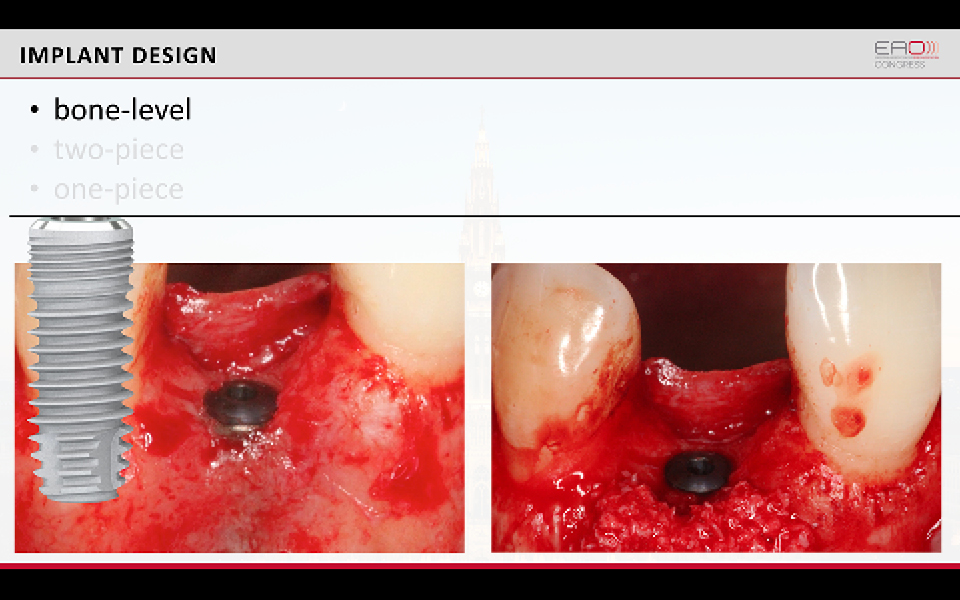
Figure 2
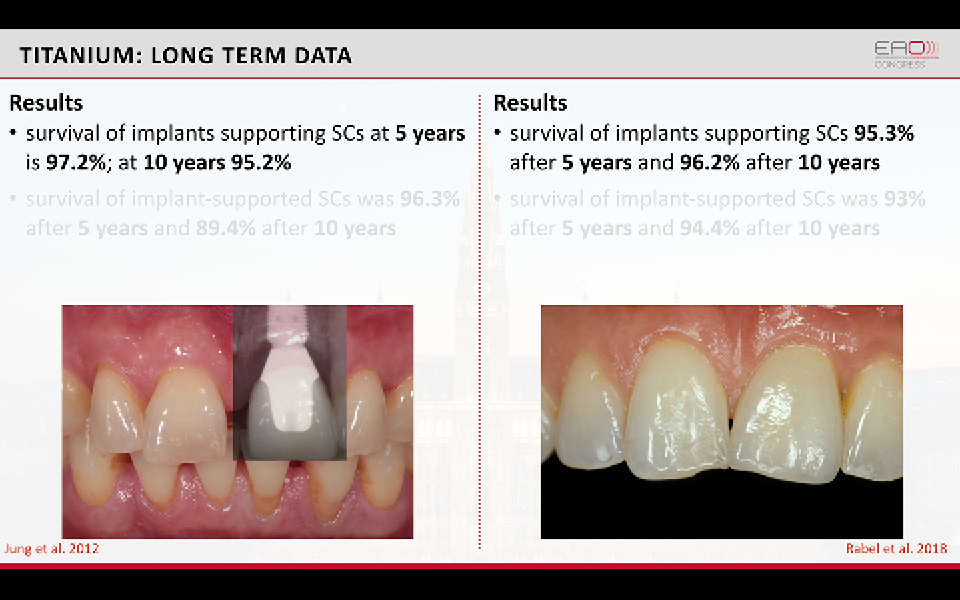
Figure 3
References:
Albrektsson T, Chrcanovic B, Mölne J, Wennerberg A. Foreign body reactions, marginal bone loss and allergies in relation to titanium implants. Eur J Oral Implantol. 2018;11 Suppl 1:S37-S46.
Buser D, Sennerby L, De Bruyn H. Modern implant dentistry based on osseointegration: 50 years of progress, current trends and open questions. Periodontol 2000. 2017 Feb;73(1):7-21. doi: 10.1111/prd.12185.
Jung RE, Zembic A, Pjetursson BE, Zwahlen M, Thoma DS. Systematic review of the survival rate and the incidence of biological, technical, and aesthetic complications of single crowns on implants reported in longitudinal studies with a mean follow-up of 5 years. Clin Oral Implants Res. 2012 Oct;23 Suppl 6:2-21. doi: 10.1111/j.1600-0501.2012.02547.x.
Mombelli A, Hashim D, Cionca N. What is the impact of titanium particles and biocorrosion on implant survival and complications? A critical review. Clin Oral Implants Res. 2018 Oct;29 Suppl 18:37-53. doi: 10.1111/clr.13305.
Rabel K, Spies BC, Pieralli S, Vach K, Kohal RJ. The clinical performance of all-ceramic implant-supported single crowns: A systematic review and meta-analysis. Clin Oral Implants Res. 2018 Oct;29 Suppl 18:196-223. doi: 10.1111/clr.13337.
Pro zirconia
Titanium implants work fairly well in most situations. What can we do better?
“No differences in osseointegration have been found between titanium and zirconia”
Various aspects of osseointegration have been studied: biomechanics has been evaluated by measuring removal torque; histological factors have been researched by looking at bone-to-implant contact in animal experiments. Results of these assessments were similar for titanium and zirconia implants (Manzano et al. 2014).
Peri-implant soft tissue has also been evaluated, and the authors of an experimental study reported similar soft tissue integration around titanium and zirconia implants (Thoma et al. 2015). In both types of implant, it was found that peri-implant mucosa was not influenced by the material. The factors which had the most influence on the soft tissue were implant design and the position of the microgap. This conclusion is in accordance with other studies on zirconia implants and with previous experimental studies on different abutment materials (Abrahamsson et al. 1998; Welander et al. 2008).
“It was found that peri-implant mucosa was not influenced by the material”
Regarding the clinical performance of zirconia implants, according to a recent meta-analysis, the one-year survival rates amount to 95.6% (Pieralli et al. 2017). An ongoing five-year follow-up study found a 98.4% survival rate in a cohort of 53 patients and 63 one-piece zirconia implants (Balmer et al. 2018.). These findings are summarised in Figure 1.
The majority of zirconia implants have been fabricated as one-piece (Figures 2 and 3) and the clinical limitations of this design are well-documented. In fact, because of their design, one-piece implants have reduced surgical versatility and prosthetic flexibility, and the removal of cement can be difficult, especially in anterior areas. Two-piece zirconia implants are now available, mostly with a tissue-level design (Figures 4 and 5).
Presentation figures

Figure 1
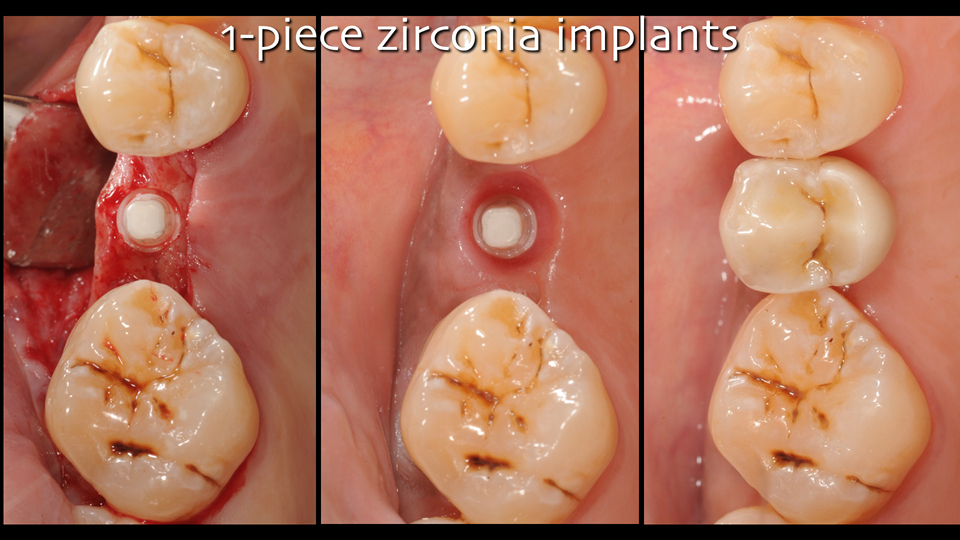
Figure 2
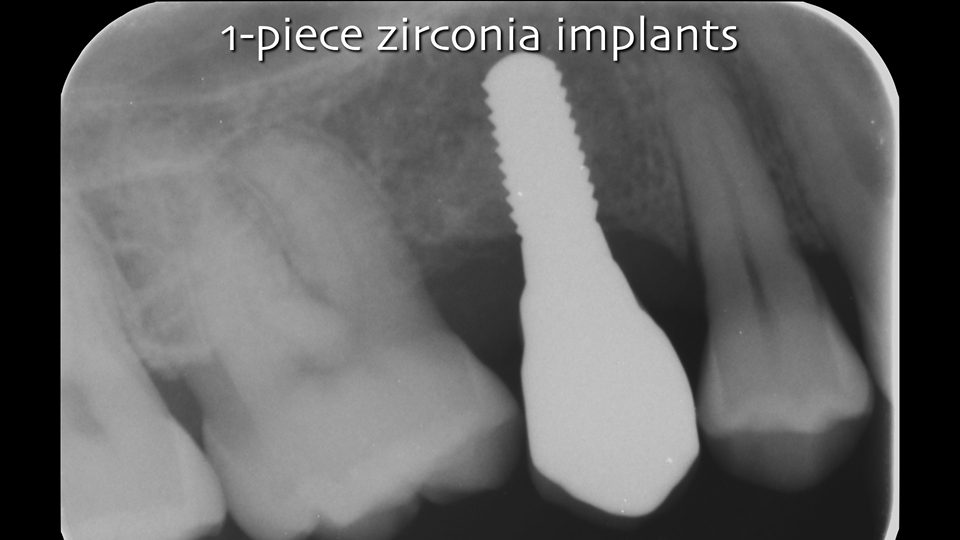
Figure 3
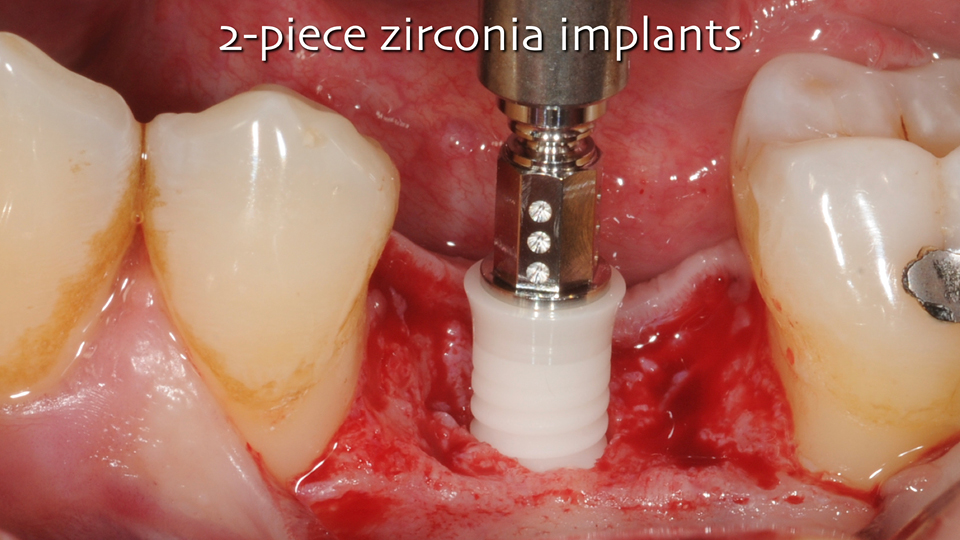
Figure 4
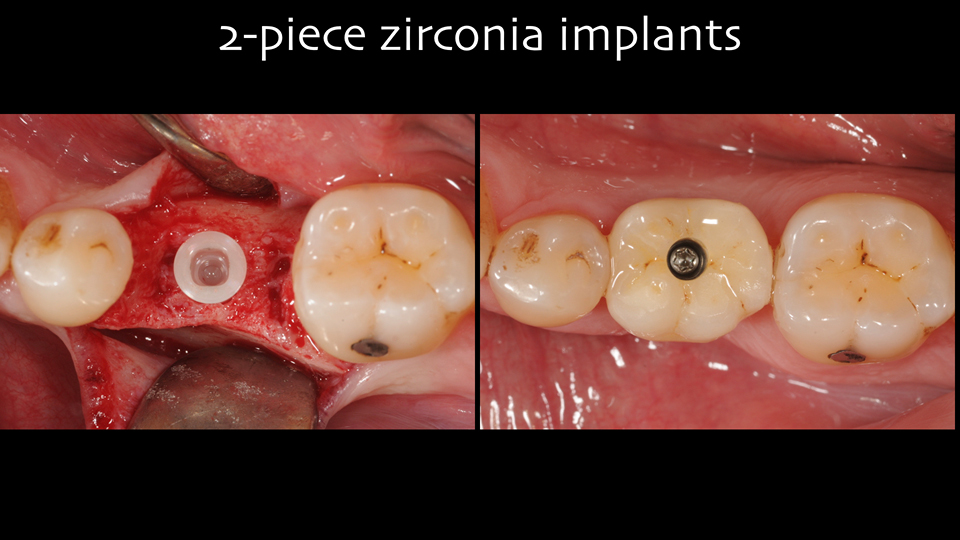
Figure 5
References:
Abrahamsson I1, Berglundh T, Glantz PO, Lindhe J. The mucosal attachment at different abutments. An experimental study in dogs. J Clin Periodontol. 1998 Sep;25(9):721-7.
Balmer, M. Zirconia implants as abutments for single crowns and fixed dental prostheses- 5 years results of a prospective cohort investigation. Abstract submitted to the AEO Meeting 2018. EAO Online Library. Balmer M. Oct 8, 2018; 234709
Manzano G, Herrero LR, Montero J. Comparison of clinical performance of zirconia implants and titanium implants in animal models: a systematic review. Int J Oral Maxillofac Implants. 2014 Mar-Apr;29(2):311-20. doi: 10.11607/jomi.2817.
Pieralli S, Kohal RJ, Jung RE, Vach K, Spies BC. Clinical Outcomes of Zirconia Dental Implants: A Systematic Review. J Dent Res. 2017 Jan;96(1):38-46. doi: 10.1177/0022034516664043. Epub 2016 Oct 1.
Thoma DS, Benic GI, Muñoz F, Kohal R, Sanz Martin I, Cantalapiedra AG, Hämmerle CH, Jung RE. Histological analysis of loaded zirconia and titanium dental implants: an experimental study in the dog mandible. J Clin Periodontol. 2015 Oct;42(10):967-75. doi: 10.1111/jcpe.12453. Epub 2015 Oct 28.
Welander M, Abrahamsson I, Berglundh T. The mucosal barrier at implant abutments of different materials. Clin Oral Implants Res. 2008 Jul;19(7):635-41. doi: 10.1111/j.1600-0501.2008.01543.x. Epub 2008 May 19.
Discussion
The reduced prosthetic flexibility of one-piece zirconia implants may pose a problem if:
- the patient’s prosthetic requirements mean that the implant needs to be re-used in a different type of reconstruction in future
- the abutment has to be reshaped and ground without damaging the chances of osseointegration. The speakers mentioned that they do grind zirconia abutments for minor adjustments and do not encounter any problems
- the restoration has to be cemented in a highly scalloped area because it could be hard to remove the cement completely
The wider uptake of zirconia implants may be hindered because of its reduced portfolio. There are no narrow zirconia implants available or solutions for tilted implants or zygomatic implants etc. Further, zirconia implants are classed as a premium product because of the high cost associated with them.
“The wider uptake of zirconia implants may be hindered because of its reduced portfolio”
When it comes to two-piece zirconia implants, the restoration cannot be completely metal-free. For mechanical reasons, titanium is used in the abutment base and the screw. It is unclear whether the interface between these two materials will pose a problem, as medium-term clinical data on this matter is still lacking. Further technical progress will allow us to overcome the current limitations of the material.
There is also currently no clinical data on the prevalence of peri-implantitis in zirconia implants. It seems that this would be related more to surface topography and implant design than material, but data to corroborate this theory is lacking.
“There is also currently no clinical data on the prevalence of peri-implantitis in zirconia implants”
Zirconia implants are mainly used as single-tooth or short-term fixed partial dentures (FPDs). These types of cases represent around 80% of cases treated in the average private European office, but there are some clinicians trying to ‘push the envelope’ who perform completely metal-free full-arch reconstructions.
The risk of fractures is a concern for zirconia implants because of the innate brittleness of ceramics. A clinical study on 170 one-piece zirconia implants (Gahlert et al. 2012) reported a fracture-rate of 10% in 3.25mm diameter implants at a mean follow-up of around three years. It is recommended that zirconia implants have a diameter of no less than 4mm in order to reduce the risk of fracture by overloading. Other related issues which clinicians should be mindful of while using zirconia implants are:
- avoid over-torqueing during implant insertion
- be careful in patients with bruxism
- avoid cantilevers, because zirconia is not as flexible as titanium
“The risk of fractures is a concern for zirconia implants because of the innate brittleness of ceramics”
The aging of zirconia because of low-temperature degradation does not represent a clinically relevant problem. More time is needed, however, to answer this question definitively.
Scientific evidence
- no difference in osseointegration: evidence type IV (pre-clinical studies)
- no difference in soft tissue integration: evidence type IV (pre-clinical studies)
- no differences in clinical performance (survival rates and marginal bone stability): evidence type I-II (RCTs), provided that the implant diameters were at least 4 mm
References:
Gahlert M, Burtscher D, Grunert I, Kniha H, Steinhauser E. Failure analysis of fractured dental zirconia implants. Clin Oral Implants Res. 2012 Mar;23(3):287-93. doi: 10.1111/j.1600-0501.2011.02206.x. Epub 2011 May 5.



