Risks and complications
Complication management
Introduction
Every clinician has the same dream: to avoid complications. But the reality is the same for us all. A growing incidence of all kinds of complications (surgical, biological and prosthetic) has been observed. This could be explained by the increased numbers of people seeking implant dentistry, and the growing number of clinicians using sub-optimal protocols.
“Every clinician has the same dream: to avoid complications”
Whatever the cause of this ‘complications overload’, it has two effects which we should bear in mind:
- it is now mandatory for every practitioner to know how to prevent complications and how to handle them if and when they occur
- clinicians must have informed the patient from the very beginning of treatment about potential problems
Surgical complications
Surgical complications are inevitable during a career in surgery. A complication rate of 2% means that 1 in every 50 patients will experience complications. For clinicians, this means a 98% success rate. But for every 50th patient, it means a 100% complication rate. The speaker recommended that we always consider such statistics from the patient’s perspective. Most complications can be prevented by controlling local and general contributing factors.
“A complication rate of 2% means that 1 in every 50 patients will experience complications”
Haemorrhaging is an immediate complication which every surgeon should be able to manage. All surgeons should:
- be familiar with anticoagulants and drug interactions
- be able to diagnose the individualised anatomy of the surgical field
- be able to prevent haemorrhage in the submandibular-sublingual space with lingual protection when drilling, since it should be considered as a life-threatening emergency
In order to reduce the risk of potential mandibular fracture in highly atrophic cases, it is important to preserve the mandibular structure; not drill through the lower border; not place too many implants; and ensure that there is adequate distance between implants.
Regarding nerve injuries, most cases reported as a consequence of implant surgery refer to mandibular nerve damage (up to 30% – a very worrying figure). The key is to act as soon as the injury is suspected, remove the implant(s) within 12–30 hours and refer the patient for further management. Another related issue is neuropathic pain, a neurosensory disturbance which is often caused by nerve injuries but sometimes arises from an obscure.
“The key is to act as soon as the injury is suspected”
Grafting complications can be summarised as:
- wound dehiscence, caused by poor technique, tension across the suture line or contamination/necrosis of the augmented material
- perforations of the Schneiderian membrane, a very frequent complication which may lead to release and dissemination of the graft material throughout the sinus and to sinusitis
- unrealistic expectations, the most common of which is what can be achieved by GBR
Whilst penetration of the sinus floor by an implant is unlikely to cause clinical signs or symptoms, in cases of reduced residual alveolar height and compromised implant stability, displacement of the implant into the sinus is a risk. Loss of an implant or grafting material into the sinus will often require ENT intervention.
Poor implant positioning is the most common complication and it is almost always caused by the surgeon, either because of lack of planning, disorientation, or inadequate skill. This can lead to long-term problems such as functional deficit, aesthetic compromise or higher risk of biological complications. Implant removal to correct these issues is rarely a simple treatment.
“Poor implant positioning is the most common complication”
The treatment of surgical complications means that the patient must undergo more surgery; the site will become even more compromised; and there will be even more risk with subsequent procedures.
The impact of a complication is far-reaching. For the patient, they are affected not only physically but also psychologically and financially. For the surgeon, they are affected psychologically, legally and financially; their reputation will also be affected. There is therefore a lot to gain by preventing complications in the first place. This requires:
- comprehensive patient assessment
- restoration-driven preparatory planning
- competent and aseptic surgical technique
Checklists are highly recommended for preventing complications. The speaker said that surgical error is the final result of several factors that coincide, and compared the situation to the holes in Swiss cheese, which come together to be aligned by chance (Figure 1).
Presentation figures

Figure 1
Biological complications
Biological complications are common in implant therapy: the prevalence of peri-implant mucositis (PM) was recently reported as 43% and peri-implantitis (PI) as 22% (Derks & Tomasi. 2015). It has been reported that patients with peri-implant mucositis, if not treated, have a six times higher risk for peri-implantitis (Costa et al. 2012).
“The prevalence of peri-implant mucositis (PM) was recently reported as 43% and peri-implantitis (PI) as 22%”
A recent systematic review concluded that mechanical plaque control should be considered the standard of care in management of PM (Salvi & Ramseier. 2015). There are a number of adjunctive mechanical/chemical methods for implant surface decontamination, but they do not seem to improve the efficacy of regular debridement. Air polishing devices are simple to use, do not make marked changes in transmucosal implant surfaces and appear equally efficient as other types of instruments used for the mechanical decontamination of implants (Schwarz et al. 2015). Although PM can usually be completely resolved, this should not be expected in all patients, and some cases may evolve into PI.
PI should be addressed surgically, since only non-surgical treatment of PI is not commonly ineffective in disease resolution. The strategic position of the implant and extent of bone loss should be considered in the decision to treat the implant surgically or to remove it. The surgical approach must take into account the morphology and extent of the defect, the type of implant surface, and local and systemic conditions (Figure 1).
“PI should be addressed surgically”
There is no general consensus on which treatment method is more effective. We should therefore rely on consensus from experts (Heitz-Mayfield et al. 2014):
- reducing risk factors is crucial, especially by controlling the patient’s oral hygiene and the design and cleansability of the prosthesis
- non-surgical debridement should be performed before any surgical intervention (for example, air-polishing with a subgingival tip)
- the site should be re-assessed and surgically treated after 3–4 weeks, when less inflammation is usually present
- the aim of surgery is to access the implant surface to decontaminate it, and there are many different combinations of mechanical and chemical methods available. No recommendations for a specific method can be given, as complete biofilm removal appears to be impossible (Ntrouka et al. 2011)
- following decontamination, the practitioner should choose between a resective or regenerative approach. The decision will depend primarily on:
- the configuration of the bony lesion
- the type of surface of the implant
Following the classification proposed by F. Schwarz (Schwarz et al. 2007; Schwarz et al. 2010), circumferential bony defects (class Ie) respond better to regenerative therapy than bony defects with some degree of buccal dehiscence (class Ib or class Ic). Horizontal defects (class II) should only be treated by a resective approach, especially in non-aesthetic areas.
A preclinical in vivo study has demonstrated that the type of implant surface affects the treatment outcome of PI (Albouy et al. 2011). This should therefore be taken into account when selecting the proper treatment.
- in cases involving horizontal defects and machined implants, a resective approach should be undertaken
- if implants with a modified surface are being used, implantoplasty should be added to the treatment plan, as it has been shown to minimise the progression of bone loss (Romeo et al. 2005; Romeo et al. 2007)
- in cases where class Ib or Ic bony defects are present, a combined approach is recommended, involving implantoplasty at the aspect of the implant harboring a dehiscence, and regeneration in the intrabony part of the defect (Schwarz. 2013) (Figures 2–3)
The use of adjunctive antibiotics seems to be beneficial in cases involving rough implants, but not in cases involving machined implants treated with a resective approach. It should also be noted that the potential benefits of adjunctive antibiotics on the long-term is not known (Caruac et al. 2017).
“The type of implant surface affects the treatment outcome of PI”
The speaker concluded by saying that bone loss progresses rapidly in peri-implantitis, and therefore treatment must be undertaken quickly to minimise the damage.
Presentation figures
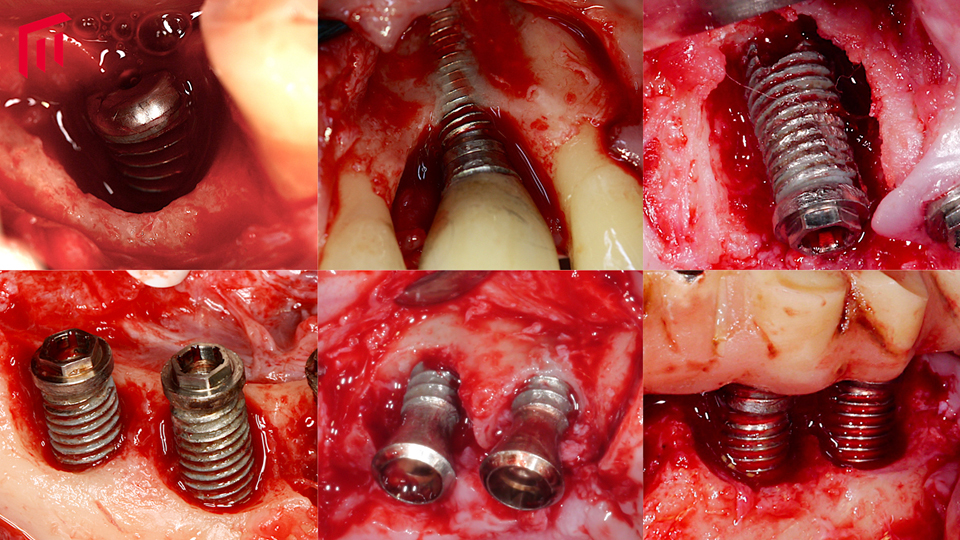
Figure 1
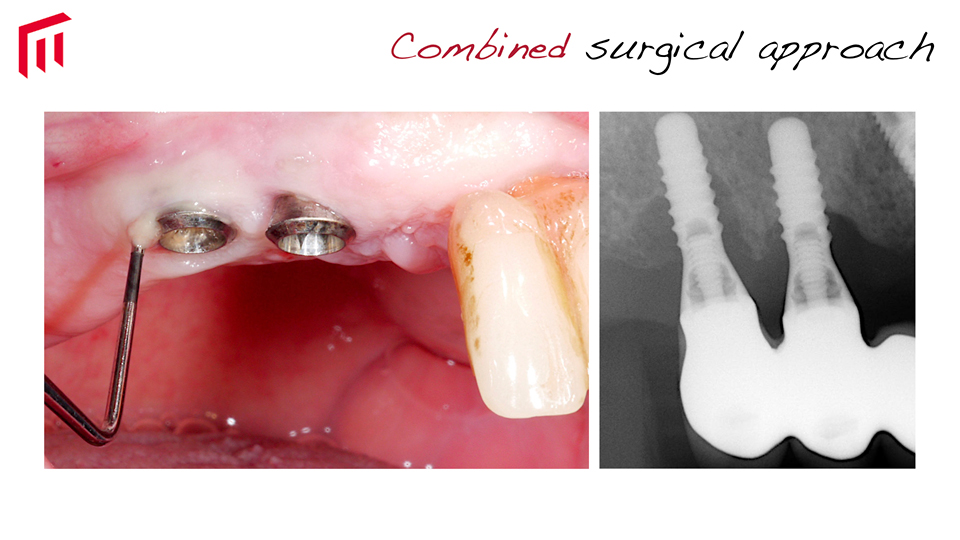
Figure 2
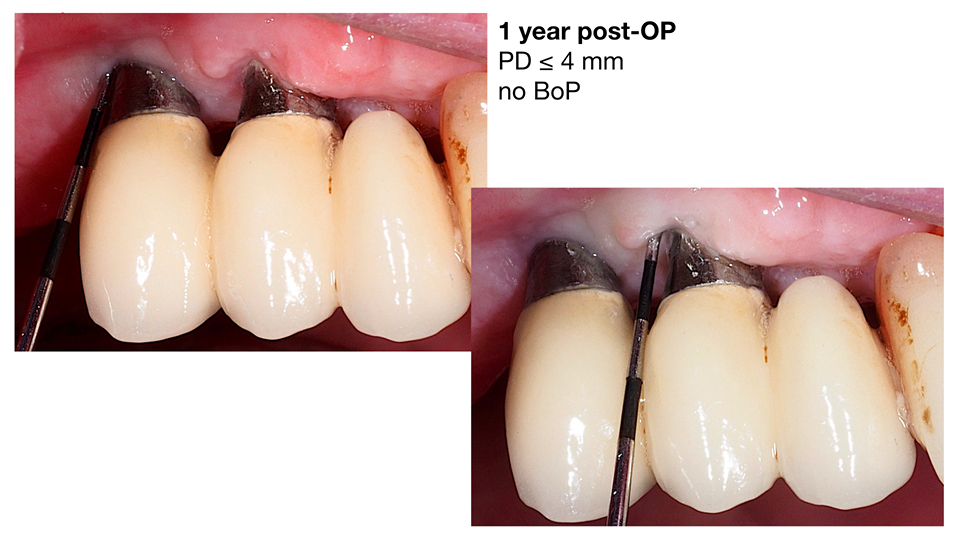
Figure 3
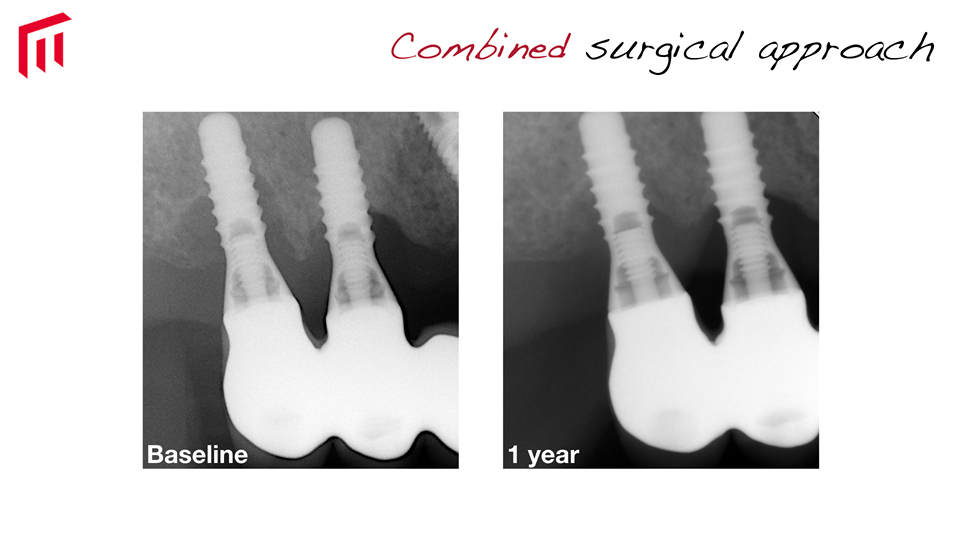
Figure 4
References:
Albouy JP, Abrahamsson I, Persson LG, Berglundh T. Implant surface characteristics influence the outcome of treatment of peri-implantitis: an experimental study in dogs. J Clin Periodontol. 2011 Jan;38(1):58-64. doi: 10.1111/j.1600-051X.2010.01631.x. Epub 2010 Nov 24.
Carcuac O, Derks J, Abrahamsson I, Wennström JL, Petzold M, Berglundh T. Surgical treatment of peri-implantitis: 3-year results from a randomized controlled clinical trial. J Clin Periodontol. 2017 Dec;44(12):1294-1303. doi: 10.1111/jcpe.12813. Epub 2017 Nov 10.
Costa FO, Takenaka-Martinez S, Cota LO, Ferreira SD, Silva GL, Costa JE. Peri-implant disease in subjects with and without preventive maintenance: a 5-year follow-up. J Clin Periodontol. 2012 Feb;39(2):173-81. doi: 10.1111/j.1600-051X.2011.01819.x. Epub 2011 Nov 23.
Derks J, Tomasi C. Peri-implant health and disease. A systematic review of current epidemiology. J Clin Periodontol. 2015 Apr;42 Suppl 16:S158-71. doi: 10.1111/jcpe.12334.
Heitz-Mayfield LJ, Needleman I, Salvi GE, Pjetursson BE. Consensus statements and clinical recommendations for prevention and management of biologic and technical implant complications. Int J Oral Maxillofac Implants. 2014;29 Suppl:346-50. doi: 10.11607/jomi.2013.g5.
Ntrouka VI, Slot DE, Louropoulou A, Van der Weijden F. The effect of chemotherapeutic agents on contaminated titanium surfaces: a systematic review. Clin Oral Implants Res. 2011 Jul;22(7):681-690. doi: 10.1111/j.1600-0501.2010.02037.x. Epub 2010 Dec 29.
Romeo E, Ghisolfi M, Murgolo N, Chiapasco M, Lops D, Vogel G. Therapy of peri-implantitis with resective surgery. A 3-year clinical trial on rough screw-shaped oral implants. Part I: clinical outcome. Clin Oral Implants Res. 2005 Feb;16(1):9-18.
Romeo E, Lops D, Chiapasco M, Ghisolfi M, Vogel G. Therapy of peri-implantitis with resective surgery. A 3-year clinical trial on rough screw-shaped oral implants. Part II: radiographic outcome. Clin Oral Implants Res. 2007 Apr;18(2):179-87.
Salvi GE, Ramseier CA. Efficacy of patient-administered mechanical and/or chemical plaque control protocols in the management of peri-implant mucositis. A systematic review. J Clin Periodontol. 2015 Apr;42 Suppl 16:S187-201. doi: 10.1111/jcpe.12321.
Schwarz F, Becker K, Renvert. Efficacy of air polishing for the non-surgical treatment of peri-implant diseases: a systematic review. J Clin Periodontol. 2015 Oct;42(10):951-9. doi: 10.1111/jcpe.12454. Epub 2015 Oct 16.
Schwarz F, Herten M, Sager M, Bieling K, Sculean A, Becker J. Comparison of naturally occurring and ligature-induced peri-implantitis bone defects in humans and dogs. Clin Oral Implants Res. 2007 Apr;18(2):161-70.
Schwarz F, Sahm N, Schwarz K, Becker J. Impact of defect configuration on the clinical outcome following surgical regenerative therapy of peri-implantitis. J Clin Periodontol. 2010 May;37(5):449-55. doi: 10.1111/j.1600-051X.2010.01540.x. Epub 2010 Mar 24.
Schwarz F, Hegewald A, John G, Sahm N, Becker J. Four-year follow-up of combined surgical therapy of advanced peri-implantitis evaluating two methods of surface decontamination. J Clin Periodontol. 2013 Oct;40(10):962-7. doi: 10.1111/jcpe.12143. Epub 2013 Aug 12.
Prosthetic technical complications
A recent epidemiological study screened almost 11,000 implants in Sweden after a mean follow-up period of 5.3 years. The study reported that technical complications requiring re-interventions by a dentist occurred in around 25% of implants (14% chipping and 11% screw loosening) (Karlsson et al. 2018). If we consider this statistic in practical terms, every clinician can expect to have to manage technical complications in roughly one in four implants.
“Every clinician can expect to have to manage technical complications in roughly one in four implants”
When managing a complication, the first step is to identify the implant system and have the appropriate tools to work with it. Next, you need to try to find out why the complication occurred. It is often the case that the restoration needs to be removed in order to properly examine the site. The patient must be informed of the risks involved when solving the technical problems.
A breakdown analysis should be performed to prevent future complications. The speaker outlined a four-step plan for this:
- Ensure that the screws are compatible and appropriately torqued
- Check the passivity between the framework and the abutments/implants
- Examine the restoration/abutment/implant interfaces on a radiograph using the parallel technique
- Verify that there is proper distribution of occlusal contacts, that there are no interferences and check whether the patient has any habits or para-functions which could be problematic
Framework misfit is more harmful in implants than in teeth. Since implant-bone interface cannot adapt to framework misfits, it can lead to bending of the framework which puts tension on the screws and the veneering porcelain.
“Framework misfit is more harmful in implants than in teeth”
The speaker explained that we should aim to minimise misfit as much as possible, since a mismatch of 150μ greatly increases the risk of fractures in the veneering material. A recent experimental study (Löfgren et al. 2017) tested misfit in partial fixed implant-supported restorations. The authors found that when screwing frameworks, gaps were no more visible in poorly-fitting frameworks than in normal ones, however the tension inside was much greater in the misfits. They found that the more dynamic cycles there were, the more cracks appeared in the porcelain. Under functional cyclic loading, framework misfits will result in complications due to fatigue. We should therefore always strive for passive fit, and should check the framework with light force.
We all know that in theory, veneering porcelain must be supported. But this is often forgotten in practice. To prevent fractures, the framework should support the veneering porcelain and this must be checked radiographically. The speaker explained that the causes for fracture and screw loosening are in fact the same.
“To prevent fractures, the framework should support the veneering porcelain and this must be checked radiographically”
Preloading ensures screw stability, but only 10% of the torqueing force translates into preload. The rest is dissipated in the friction between components, and this effect is magnified in misfits. The more friction, the more resistance needs to be overcome by the torqueing force and ultimately less preloading reaches the screw.
The amount of torque should correlate with the exact amount of combined friction the implant, abutment and screw have together. No more, no less.
There are a number of copycat materials available which claim to be compatible with abutments and screws. However, these have been shown to present critical differences in contact areas from the originals (Mattheos et al. 2015). Consequently, the fit and friction are not the same and accordingly the required torque is not either. The speaker said that we should be wary of using these copycat abutments and screws.
“We should be wary of using copycat abutments and screws”
The speaker also said that when trying to retrieve broken components, it is crucial to read the manufacturers’ manual and follow their instructions regarding which tools to use. The corresponding component retrieval kit should be used, recommended steps followed diligently and the correct magnification used.
Presentation figures

Figure 1

Figure 2
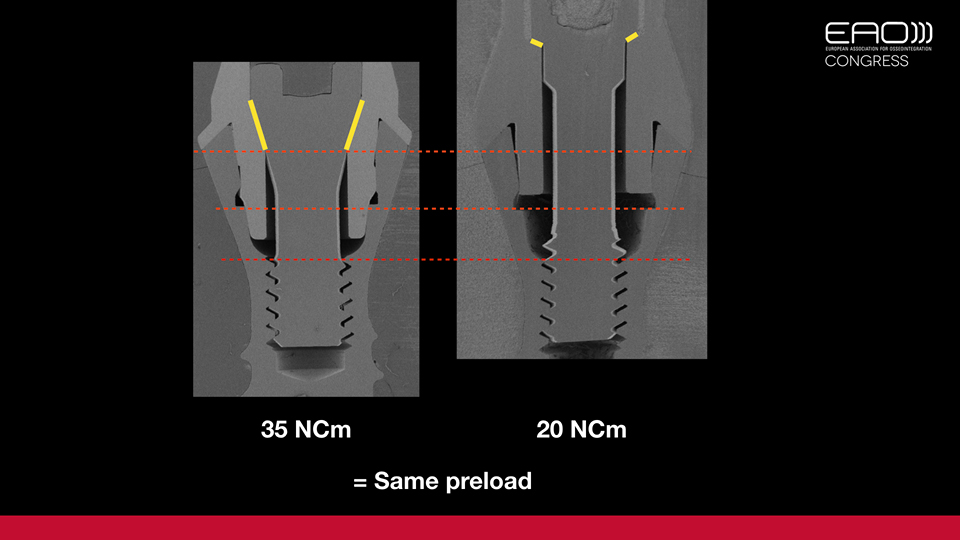
Figure 3
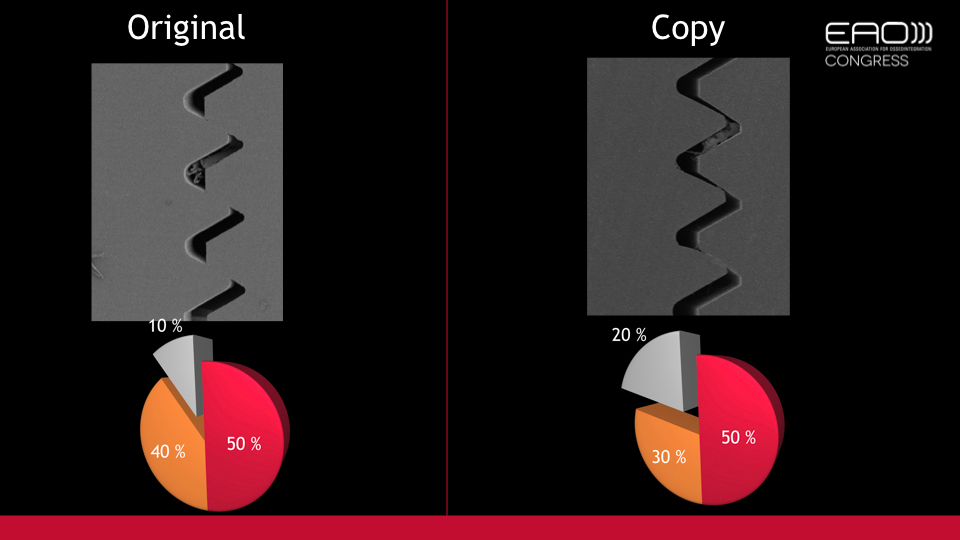
Figure 4
References:
Karlsson K, Derks J, Håkansson J, Wennström JL, Molin Thorén M, Petzold M, Berglundh T. Technical complications following implant-supported restorative therapy performed in Sweden. Clin Oral Implants Res. 2018 Jun;29(6):603-611. doi: 10.1111/clr.13271. Epub 2018 Jun 20.
Löfgren N, Larsson C, Mattheos N, Janda M. Influence of misfit on the occurrence of veneering porcelain fractures (chipping) in implant-supported metal-ceramic fixed dental prostheses: an in vitro pilot trial. Clin Oral Implants Res. 2017 Nov;28(11):1381-1387. doi: 10.1111/clr.12997. Epub 2016 Dec 19.
Mattheos N, Li X, Zampelis A, Ma L, Janda M. Investigating the micromorphological differences of the implant-abutment junction and their clinical implications: a pilot study. Clin Oral Implants Res. 2016 Nov;27(11):e134-e143. doi: 10.1111/clr.12578. Epub 2015 Mar 9.
Discussion
One of the more frequent causes of complications is poor implant positioning. At what point should we decide to remove the implant?
When malposition is suspected during implant placement, the best thing to do is remove it immediately. The difficulty and biological cost of removing an implant once osseointegration has occurred are very high. To remove a poorly positioned implant we should first try to reverse the torque (using the appropriate instrument kit for the implant brand). Generally speaking, this manoeuvre has a success rate of around 25%. In the other 75% of cases, we need to raise a flap and drill the bone at the implant side in the most conservative way possible. Trephine is not recommended as it allows more bone to become damaged and often necrotic through overheating. Regardless, once the implant has been removed, it is recommended that a healing period of five weeks is observed before placing a new one in the proper position.
To prevent peri-implantitis, would a polished transmucosal neck be beneficial?
A polished neck is only one of multiple factors that may affect the risk of peri-implantitis. In theory, a polished surface is easier to clean and should have less plaque accumulation, but there are some implants which have a polished neck but still encounter peri-implantitis. Other factors must be considered. If the trans-gingival area is not healthy, or the implant is placed too deep, or there is a sub-optimal connection, then peri-implantitis is also going to occur.
Would zirconia implants be less prone to peri-implantitis?
It is been claimed that zirconia promotes less biofilm adherence in vitro, but we cannot conclude from this that there is a lower incidence of peri-implantitis in zirconia implants. There is still limited clinical data on zirconia implants, so we really cannot give a definitive answer.
Zirconia implants are still associated with peri-implantitis, and do not have better results than titanium implants. Apart from implantoplasty – which is not possible with zirconia implants – the treatment approach is the same as when working titanium.
Is it feasible to think that an anti-infection implant surface will be available in the future?
It is a nice dream. There is lot of active research being carried out in this direction and it is possible that positive results will be available in the near future.
Are all surface decontamination methods comparable?
The most important thing to bear in mind is that low-tech methods which are simpler and cheaper work in a similar way to the sophisticated and expensive high-tech techniques (like lasers). The results are indeed comparable.
Is the incidence of chipping in zirconia restorations similar to metal-ceramic restorations?
Zirconia reconstructions are still in a development phase. The incidence of chipping on zirconia is similar to (or even greater than) metal-ceramic reconstructions. Thus, for cases involving full-arch reconstruction, the speaker said they prefer to use metal-ceramic restorations.
Does framework misfit have any influence over osseointegration?
Finite element models show force transmission between bone/implant interfaces. The results can be applied to the technical field, but it is hard to draw conclusions from these in relation to bone. However, it is reasonable to hypothesise that cortical bone can be lost from an intense concentration of forces and that the modified implant surface may be exposed to the oral environment, predisposing it to peri-implant disease. But this cannot be attributed to misfit alone.



